Case report
Transcranial direct current stimulation over the primary motor cortex and dorsolateral prefrontal cortex for Achilles’ tendon pain modulation: a case report of an elite soccer player
Luciane Aparecida Moscaleski1 (https://orcid.org/0000-0002-3746-5691)
Pedro Augusto Pontin2 (https://orcid.org/0000-0001-9667-0006)
André Fukunishi Yamada3 (https://orcid.org/0000-0001-7546-8757)
Gisele Maria da Silva4 (https://orcid.org/0000-0002-3008-1062)
Daniel Soares Gonçalves5 (https://orcid.org/0009-0001-7708-7437)
Lucas Filgueiras Freitas6 (https://orcid.org/0009-0003-2570-7467)
Gilberto Amado Rodrigues da Cunha Filho7 (https://orcid.org/0000-0001-5538-0176)
Victor Broglio Soraggi8 (https://orcid.org/0009-0009-2749-3355)
Vinicius Felice Ponzio9 (https://orcid.org/0009-0004-7137-2589)
Alexandre Moreira10 (https://orcid.org/0000-0002-4609-1839)
1Sociedade Esportiva Palmeiras (São Paulo). São Paulo, Brazil. Universidade de São Paulo (São Paulo). São Paulo, Brazil.
2-9Sociedade Esportiva Palmeiras (São Paulo). São Paulo, Brazil.
10Corresponding contact. Universidade de São Paulo (São Paulo). São Paulo, Brazil. [email protected]
ABSTRACT | INTRODUCTION: Managing partial Achilles’ tendon tears in elite soccer players during a competitive season requires a balance between structural healing and performance maintenance. Transcranial direct current stimulation (tDCS) has emerged as a potential neuromodulatory adjunct to manage pain and optimize motor efficiency through top-down regulation. OBJECTIVES: This case report evaluates the effects of a 17-session dual-site tDCS protocol on pain modulation, competitive performance, and structural tendon repair in a professional top-level soccer player. MATERIALS AND METHODS: The intervention consisted of 17 tDCS sessions (2mA) using a dual-target montage: anodal stimulation over the left primary motor cortex (M1) and left dorsolateral prefrontal cortex (DLPFC), with cathodal electrodes over the supraorbital and contralateral frontal midline areas. Assessments included the Visual Analog Scale (VAS) for pain and the Rating of Perceived Exertion (RPE) for recovery training and competitive matches. Structural evolution was monitored via serial ultrasonography (US) and confirmed by multi-sequence magnetic resonance imaging (MRI). RESULTS: The athlete exhibited a positive pain-exertion dissociation: a consistent progressive reduction in VAS pain scores occurred simultaneously with an increasing trend in match-play RPE, indicating a higher capacity for physical exertion despite the injury. Training RPE remained stable and low (1–3), reflecting controlled recovery workloads. Serial axial US initially identified a partial tear in the lateral and deep fibers with laminar fluid. A 30-day follow-up US and MRI (T1, T2-weighted with fat saturation) confirmed successful tissue repair, with intermediate-signal intensity tissue filling the defect, indicating organized scar formation without transfixing lesions. Serial axial ultrasonography initially identified a partial tear within the lateral and deep fibers of the tendon midbody, accompanied by localized laminar fluid. A 30-day follow-up, utilizing US and multi-sequence MRI (T1; T2-weighted with fat saturation), confirmed structural restoration. The previous defect was replaced by tissue of intermediate-signal intensity, consistent with organized fibroblastic repair and scar formation, in the absence of residual transfixing lesions. CONCLUSION: The dual-site tDCS intervention was associated with significant pain modulation and improved motor efficiency, allowing the athlete to increase competitive output during the structural healing phase. The correlation between reduced pain (VAS), increased match effort (RPE), and confirmed biological repair suggests that tDCS facilitated an optimal environment for recovery. This suggests that tDCS is a promising adjunct for maintaining elite performance during injury rehabilitation, though further controlled research is needed to elucidate the specific mechanisms of tDCS-mediated tissue repair.
KEYWORDS: Non-Invasive Brain Stimulation. Tendinopathy. Achilles Tendon. Team Sports. Magnetic Resonance Imaging.
How to cite this article: Moscaleski LA, Pontin PA, Yamada AF, Silva GM, Gonçalves DS, Freitas LF, et al. Transcranial direct current stimulation over the primary motor cortex and dorsolateral prefrontal cortex for Achilles’ tendon pain modulation: a case report of an elite soccer player. Brain Imaging Stimul. 2026;5:e6696. https://doi.org/10.17267/2965-3738bis.2026.e6696
Submitted Jan. 13, 2026, Accepted Mar. 19, 2026, Published 30 abr. 2026
Brain Imaging Stimul., Salvador, 2026;5:e6696
https://doi.org/10.17267/2965-3738bis.2026.e6696
ISSN: 2965-3738
Assigned editors: Fuad Hazime, Abrahão Baptista
1. Introduction
Transcranial direct current stimulation (tDCS) is a non-invasive neuromodulatory technique that utilizes a weak direct current passed through the cerebral cortex via scalp electrodes to induce polarity-dependent modulation of cortical excitability1. tDCS is typically applied via two surface electrodes, one acting as the anode and the other as the cathode. Anodal tDCS (a-tDCS) (involving applying an anode over the target area) typically has an excitatory effect on the underlying cerebral cortex by depolarizing neurons, while cathodal tDCS (c-tDCS) (involving applying a cathode over the target area) decreases cortical excitability by inducing hyperpolarization2.
In healthy subjects, tDCS effects have been demonstrated on motor evoked potentials2, cerebral blood oxygen levels measured with functional magnetic resonance imaging3, and cerebral blood flow4. Additionally, substantial evidence highlights the potential of tDCS not only to modulate neuronal components but also to significantly influence adjacent non-neuronal structures5. This evidence indicates that beyond classical neuronal polarization, the tDCS’s capacity to influence the neurovascular unit, including astrocytes, perivascular nerves, and endothelial lining. Animal models have demonstrated that tDCS can enhance cortical microvasculature blood flow and microglial activity, suggesting a complex influence on the neurovascular microenvironment that extends far beyond simple electrical shifts6. These findings reveal the functional role of various neurovascular unit cells in response to electrical stimulation, contributing to a more comprehensive explanation of tDCS effects beyond classical neuronal polarization. Furthermore, they expand the understanding of the biological impacts of tDCS, suggesting a broader and more complex influence on the neurovascular microenvironment than previously considered.
The effects of tDCS as a neuromodulatory intervention in elite sports have been investigated, and its potential has been demonstrated across different sports modalities and distinct purposes7-17. Furthermore, other effects include enhanced conditioned pain modulation, modulation of the mechanical pain threshold18, and pain perception to electrical stimulation19. Studies have shown its efficacy in enhancing conditioned pain modulation and increasing mechanical pain thresholds, primarily through stimulation of the primary motor cortex (M1) and the dorsolateral prefrontal cortex (DLPFC). While M1 is central to modulating overall cerebral excitability and sensory processing, the DLPFC plays a critical role in the cognitive and emotional dimensions of pain and pain tolerance.
Regarding the use of tDCS to modulate pain, several studies have applied tDCS over the primary motor cortex (M1)4,19,20, which modulates various different types of cortical pain processing19,20. Other tDCS studies have examined the effect of the dorsolateral prefrontal cortex (DLPFC)19,21,22 due to its role in the cognitive and emotional aspects of pain19. However, M1 stimulation has been proposed to be the main region for modulating cerebral excitability23. Indeed, the DLPFC plays a central role in pain processing24. DLPFC stimulation can play a role in regulating pain tolerance25 and also in increasing the mechanical sensitivity threshold and the pain pressure threshold23.
Despite the growing body of literature supporting tDCS in controlled settings, a significant gap remains regarding its longitudinal application in "real-world" elite sports environments. Most existing research is limited to isolated laboratory sessions or short-term rehabilitation protocols, leaving the cumulative effects and feasibility of tDCS during an active competitive season largely unexplored. Furthermore, while simultaneous stimulation of M1 and DLPFC has shown potential in clinical pain management, its integration into the daily routine of a professional athlete facing the physiological and psychological demands of high-level competition is not yet documented.
Therefore, this report aims to address this knowledge gap by evaluating the effects of a multi-session tDCS intervention (17 sessions) applied simultaneously over both M1 and DLPFC, following an adapted protocol used by Vaseghi et al.23. Specifically, the study aims to assess the impact of this intervention, combined with standard rehabilitation procedures, on the pain modulation of a professional top-level soccer player during an official competitive season. By documenting its use during active participation in high-level games rather than in an isolated setting, this report provides unique insights into the practical utility and efficacy of tDCS in the elite sports landscape.
2. Methods
2.1 Participant
One elite male professional soccer player participated in this tDCS intervention protocol. The analyzed player belonged to a team ranked 1st in the Brazilian Championship and qualified for the CONMEBOL Libertadores Semifinal and Final (2025) during the intervention period. The player is classified as an "elite athlete" due to a training volume of >10 hours per week and participation in national and international competitions26. The player is a 32-year-old male professional soccer player (height: 1.75 m; mass: 70 kg) currently competing in the Brazilian Série A. With a professional career spanning 15 years, the subject has competed at the highest international tiers, including the Italian Serie A, English Premier League, and Portuguese Primeira Liga. His professional honors include an Olympic Gold Medal, a Copa Libertadores title, and a Supercoppa Italiana.
The athlete was a midfielder presenting with chronic bilateral Achilles tendinopathy resulting from mechanical overload, with no history of local acute trauma. At the time of the intervention, the athlete exhibited localized pain and a sensation of stiffness associated with bilateral thickening of the Achilles tendon substance. Despite these symptoms, there was no clinical evidence of joint instability, loss of muscle strength, or reduced range of motion. Notably, the athlete did not cease his professional activities; he maintained full participation in the team’s high-performance training program and official competitions throughout the treatment period.
Marking a novel approach within the institution, tDCS was introduced for the first time at this club specifically for the treatment of Achilles tendinopathy. This protocol was integrated into the habitual multimodal rehabilitation program adopted by the medical staff. Before the initiation of the tDCS protocol, the athlete underwent a preparatory phase consisting of Focused Shockwave Therapy (BTL 6000), involving 2 sessions (2000 impulses per session, energy flux density of 0.30 mJ/mm², frequency of 10 Hz) applied directly posterior to the Achilles tendon.
During the experimental period, the tDCS intervention comprised 17 application sessions. Throughout this timeframe, the athlete continued a daily comprehensive and multimodal rehabilitation program managed by the club's medical staff, which included: Physical Agents (electrotherapy, thermotherapy, and phototherapy); Exercise-Based Therapy (kinesiotherapy, hydrotherapy, and analytical/functional strengthening); and Performance Integration (biomechanical adjustments and controlled transition phases to field-specific demands). The player received detailed instructions regarding the intervention protocol.
2.2 Experimental design
Upon reporting to the training center for his daily routine, the athlete was taken to the evaluation room for the imaging examination (ultrasound), aimed at analyzing the Achilles’ tendon. Achilles’ tendon assessments were performed using the Aplio i800 ultrasonography equipment (Canon Medical Systems, Japan). This is a high-resolution imaging system featuring beamforming technology and multifrequency transducers, allowing for defined edges, tissue contrast, and detailed analysis of the muscle-tendon architecture. The athlete was evaluated after all match sessions in a controlled environment, utilizing the same operator and anatomical positioning protocol. Images were obtained with the i24LX8 linear transducer, operating at a frequency between 7 and 18 MHz, ensuring the standardization and precision of the measurements.
Subsequently, the athlete completed the subjective scales: the 10-point Visual Analog Scale (VAS) for Pain, where 0 represented absence of pain and 10 represented unbearable pain27; and the Subjective Rating of Perceived Exertion (RPE) Scale, based on the adapted Borg CR-10 version by Foster et al.28, referring to the training and the match from the previous day. After completing the scales, the athlete was positioned in a reclining chair to ensure physical comfort and postural stability during the tDCS session. The stimulation was administered in strict accordance with the established experimental protocol parameters. The intervention was approved by the club and the Research Ethics Committee of the University (CAAE: 67915323.4.0000.5391). All procedures involving human participants were conducted in accordance with the 1964 Declaration of Helsinki.
Figure 1. Study Flow Diagram
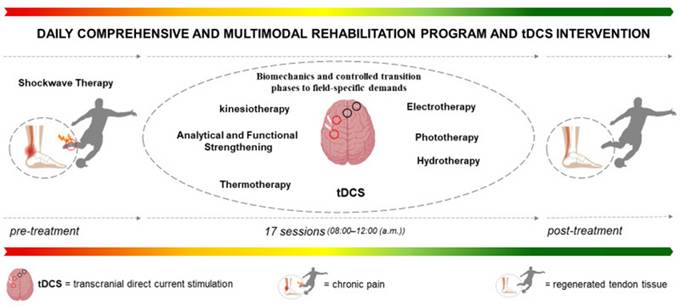
The flowchart details the intervention sequence, starting from the baseline diagnosis, followed by the preparatory focused shockwave therapy and the subsequent experimental phase consisting of 17 tDCS sessions integrated with the club's multimodal rehabilitation program.
2.3 Transcranial direct current stimulation (tDCS)
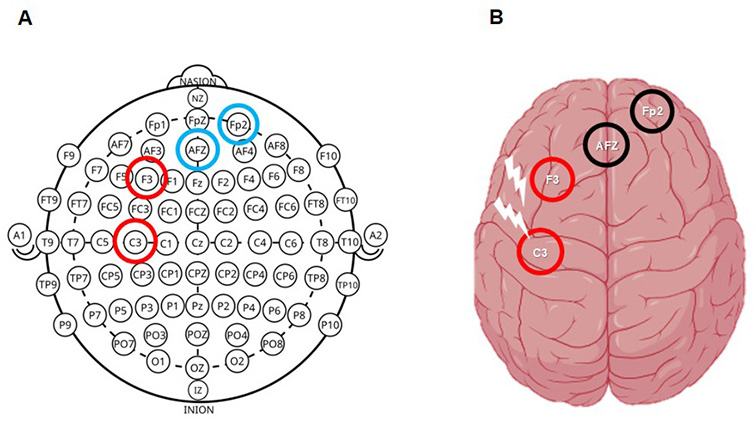
Electrode placement followed the international 10/10 EEG system model, an extended version of the international 10–20 EEG system, which provides a more specific scalp distribution29,30. The athlete's head circumference measurement was used to determine the correct size and positioning of the cap. The tDCS protocol was administered using a total constant current of 2mA for 20 minutes, including a 30-second ramp-up and ramp-down period. The stimulation was delivered via a battery-driven constant electric stimulator (MicroEstim – Genius, NKL®, Brusque, SC, Brazil). The total current was distributed through a 'Y' splitter cable to two active anodal electrodes (1mA per electrode). The anodes were positioned over the left dorsolateral prefrontal cortex (DLPFC) and the left primary motor cortex (M1), while two cathodal electrodes were placed over the supraorbital area and the contralateral frontal midline (Figure 2). The electrodes consisted of circular conductive silicone (42 mm diameter, surface area, approximately 13.85cm2) inserted into saline-soaked sponge pads (30cm2) resulting in a current density of 0.033mA/cm2 per stimulation site. To ensure the highest level of safety and procedural consistency, all sessions were conducted by a trained professional with over nine years of experience in tDCS application.
Figure 2. Electrode positioning and cortical mapping
A) Schematic representation of the international 10/10 system for EEG electrode placement in the present report (red = anodal; blue = cathodal). Cap for 128 channels. B) Cortical projection of selected electrodes (F3, C3, AFz, and Fp2). (red = anodal; black = cathodal).
3. Results
3.1 Description of changes associated with the athlete's behavior
The player underwent two sessions of focused shockwave therapy on the Achilles tendon before the start of the tDCS sessions, aiming to reduce pain and promote local tissue recovery. Prior to the tDCS intervention, the athlete’s pharmacological management for pain consisted of the regular oral intake of a fixed-dose combination (50mg Diclofenac Sodium, 300mg Paracetamol, 125mg Carisoprodol, and 30mg Caffeine; commercially known as Tandrilax®), consuming three tablets before each match. Additionally, the athlete frequently required pre-game analgesia via intramuscular injection of Ketoprofen (100 mg in a 2 mL solution, single dose; commercially known as Profenid®) to manage acute pain episodes. Following the initiation of the experimental tDCS protocol, the athlete spontaneously reduced the oral intake to a single tablet per match and completely ceased the use of injectable Ketoprofen, reporting significant clinical improvement and the ability to compete without the need for systemic anti-inflammatory injections.
Regarding functional capacity, the athlete was consistently available for selection and capable of completing the full 90 minutes of play throughout the intervention period. Any instances where the athlete did not complete a full match were exclusively due to technical or tactical substitutions by the coaching staff, rather than physical limitations or pain-related impairment in the Achilles’ tendon.
Importantly, throughout the tDCS sessions, the athlete reported a complete absence of pain in the Achilles tendon region during the first 25 minutes of the matches, a period in which he previously experienced intense discomfort. Prior to the intervention (adding tDCS to the habitual rehabilitation program), the pain typically intensified after this initial period, reaching a peak that limited him to maintain performance. However, following the introduction of tDCS, the athlete reported that when pain did emerge in the later stages of the match, it was of significantly lower intensity and did not limit his functional capacity. The player noted that his running became more fluid and less restricted, feeling able to 'give more' on the field. During this same period, the athlete scored his first three goals in the competition, attributing this performance to the absence of acute pain and improved movement fluidity. It is noteworthy that the athlete maintained his usual physiotherapy treatment and regular training routine without changes in volume or intensity.
3.2 Pain perception and effort perception
Figure 3 illustrates the progression of the player's subjective measures during the tDCS intervention. A notable trend was observed in the match Rate of Perceived Exertion (RPE). This suggests that the athlete was able to sustain higher physical demands and greater involvement in official matches as the protocol progressed. Crucially, despite this increase in match intensity, the Visual Analog Scale (VAS) for pain did not follow the same upward trend; instead, it remained stable or decreased compared to the initial sessions. This decoupling between exertion and pain intensity indicates improved functional tolerance. Regarding training RPE, values remained predominantly low (between 1 and 3), reflecting the recovery-focused nature of mid-week sessions, with a single high-intensity field session that was well-tolerated by the athlete.
Figure 3. Evolution of Pain Perception (VAS), Rating of Perceived Exertion (RPE), and training
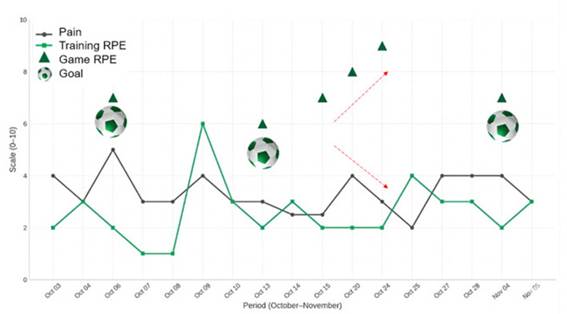
This time-series plot shows the progression of the player's subjective measures over 17 sessions (October 3rd to November 5th); PAIN (VAS) (Black Line): Tracks the visual analog scale score (0-10); Training RPE (Green Line): Tracks the subjective perceived exertion for training (CR-10 scale); Match RPE (Green Triangles): Tracks the subjective perceived exertion for matches (CR-10 scale); Goal (Soccer Ball Icon): Indicates when the athlete scored a goal.
Throughout the tDCS sessions, Achilles’ tendon ultrasonography was performed, as shown in figure 4, in the axial plane. It revealed a partial tear involving the lateral and deep fibers in the tendon body, with the presence of interposed laminar fluid, and no evidence of a transfixing lesion. The control examination, performed after 30 days, showed the partial tear area filled by scar tissue, indicating evolution of the tissue repair process.
Figure 4. Serial Axial Ultrasonography of the Achilles’ Tendon
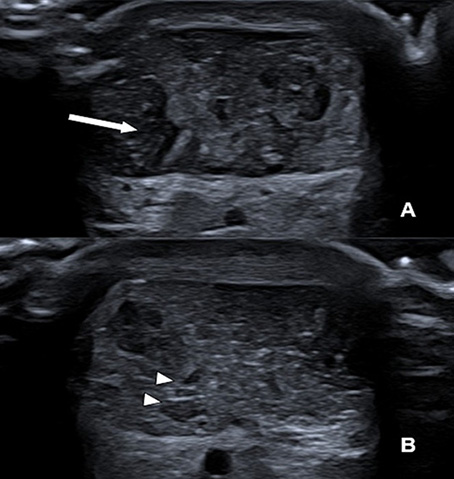
Initial Assessment (A): Axial view of the mid-portion of the Achilles tendon showing a focal area of hypoechogenicity involving the lateral and deep fibers, consistent with a partial-thickness tear. Note the presence of thin, anechoic laminar fluid (arrow) within the defect, with no evidence of complete fiber discontinuity. 30-Day Follow-up (B): The follow-up scan demonstrates significant structural remodeling. The previously identified hypoechoic defect is now filled with more organized, echogenic tissue, representing the formation of organized scar tissue and the resolution of the peritendinous fluid.
In the Achilles’ tendon magnetic resonance imaging (MRI) (Figure 5), axial T2-weighted sequences with fat saturation, axial T1-weighted sequences, and sagittal T2-weighted sequences with fat saturation identified intermediate signal tissue filling the area corresponding to the partial tear in the tendon body, confirming the structural finding previously observed.
Figure 5. Magnetic resonance imaging of the Achilles’ tendon
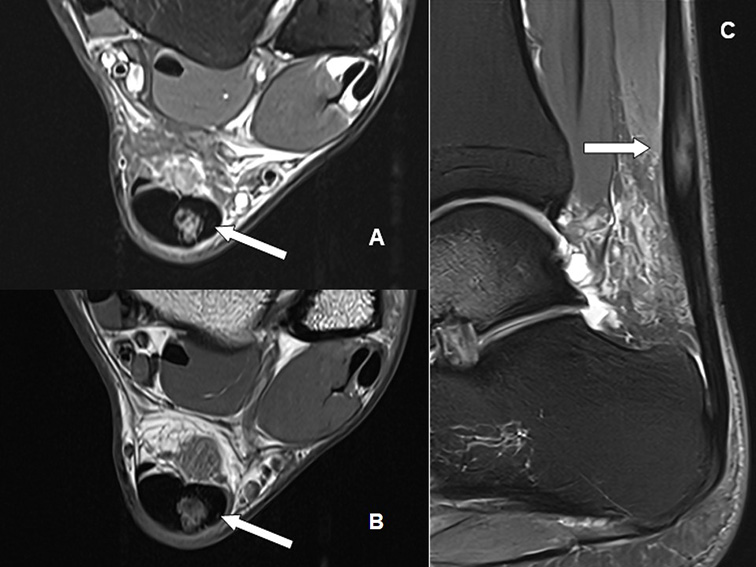
Axial T2-weighted sequence with fat saturation (A); axial T1-weighted sequence (B), and sagittal T2-weighted sequence with fat saturation (C). Note the intermediate signal tissue filling the partial tear area in the Achilles tendon body (white arrows).
4. Discussion
The primary objective of this case report was to evaluate the clinical and structural effects of a dual-site tDCS protocol in an elite professional soccer player managing a partial Achilles tendon tear during a competitive season. The main findings of this intervention demonstrate a successful clinical trajectory, characterized by a positive Pain-Exertion dissociation, as a consistent reduction in VAS pain scores occurred simultaneously with an increasing trend in match-play RPE, indicating that the athlete was able to elevate competitive physical output while experiencing diminishing pain levels. The athlete reported improved movement fluidity and the absence of pain during the initial phases of matches. Indeed, serial axial ultrasonography and multi-sequence MRI (T1 and T2-weighted fat saturation) confirmed that symptomatic improvement was matched by biological healing. The initial partial tear and laminar fluid were replaced by organized, intermediate-signal intensity scar tissue, indicating successful tissue repair without the development of transfixing lesions.
Regarding the modulation of pain perception, it is important to highlight that the sensory and emotional processing of pain involves parallel brain structures31. Lateral thalamic nuclei and the somatosensory cortex (S1) are considered responsible for the sensory-discriminative aspects of pain, such as threshold, quality, location, and intensity assessment, while medial thalamic nuclei, the prefrontal cortex, and the limbic system are considered responsible for the affective-emotional dimension of pain. The overlap between these areas and the brain regions involved in emotional processing may explain the subjective qualities of pain in humans32 and may aid in the explanation of the reduction of the perceived pain by the assessed player.
In the sports setting, Gonçalves et al.9 recently demonstrated a reduction in perceived muscle pain on the second day following official soccer match when tDCS over the DLPFC was applied as a recovery strategy. The findings suggest that tDCS can modulate the perception of recovery and sleep quality, while also influencing physiological markers like creatine kinase (CK). While Gonçalves et al. focused on acute post-match recovery, Nascimento et al.33 demonstrated that combining tDCS with peripheral stimulation is superior to using either intervention alone for pain reduction in chronic conditions. This synergistic effect likely explains why the integration of tDCS into the club’s habitual multimodal program resulted in such significant clinical and functional gains. Moreover, the functional improvement observed in the investigated athlete, reporting more 'fluid' movement, might also be linked to the cognitive benefits of tDCS. Beyond pain modulation, for example, earlier, Vitor-Costa et al.34 demonstrated that tDCS can improve the perception of effort and performance by modulating the activity of brain areas related to exercise tolerance. This is consistent with our findings, where the athlete's match RPE increased significantly without a proportional increase in pain, suggesting a higher threshold for both physical and cognitive demands.
Furthermore, the reduction in discomfort in the initial 25 minutes of play likely reduced the 'cognitive load' associated with pain monitoring, allowing the athlete to dedicate more attentional resources to tactical decision-making and technical execution, which might explain the improved clinical and competitive outcomes. Brain mapping studies have consistently identified the brain areas that are activated when someone feels pain35. These areas are mostly multimodal and respond to relevant non-noxious (non-nociceptive) stimuli, as well as noxious (nociceptive) stimuli36. The brain areas involved in the processing of pain signals that are also superficial to the skull include the primary sensory cortex (S1), the primary motor cortex (M1), and the dorsolateral prefrontal cortex (DLPFC)37. The S1 area, with its topographical organization, was long considered a key site of pain-related brain activity. However, evidence supporting this notion remains controversial. Some studies clearly show that S1 activity is related to pain intensity38,39, while others do not demonstrate such a relationship40,41. Indeed, activation of the M1 may affect pain reduction not only due to existing neural connections between the primary somatosensory cortex (S1) and M1 but also due to the functional relationship between M1 and the thalamus42. Thalamus activation leads to the activation of other pain-related structures, such as the anterior cingulate cortex and the periaqueductal gray matter, which play an important role in pain control43.
Moreover, the DLPFC is one of the most commonly activated brain areas during pain44. Changes in connectivity between the DLPFC and deeper pain-related areas45, as well as a reduction in gray matter density and DLPFC volume, have been implicated in chronic pain, presenting an alternative outcome. DLPFC activation appears to be related to the cognitive and attentional processing of noxious stimuli32 and likely plays a role in modulating pain expectation46 and pain-induced anxiety47. In tandem, these mechanisms may explain the observed results in the present case study and could be considered to interpret the results of the treatment based on the application of the present tDCS montage (M1 and DLPFC).
tDCS is a common NIBS method for modulating the cortical activity of superficial areas relevant to pain; it has been used to treat a variety of clinical conditions and is a painless technique with minimal side effects48. The proposed mechanism behind the immediate effects of tDCS is the polarity-dependent alteration of the resting membrane potential and the consequent change in corticospinal excitability at the stimulation site. The idea is that this alteration leads to the facilitation or inhibition of superficial structures and deeper, remote brain areas related to pain modulation4. Additionally, the long-lasting effects of tDCS depend on changes in N-methyl-D-aspartate (NMDA) receptor efficacy49. NMDA receptor involvement induces neuroplasticity, in which the transformation of synaptic strength occurs through long-term potentiation and depression (LTP and LTD) mechanisms2.
Thus, the effects of tDCS in the present case study might be at least in part attributed to changes in top-down regulatory mechanism, related to cortical inhibitory control and dependent on the projection of fibers from the motor cortex to structures involved in pain processing. The changes of the player’s pain perception in parallel with improved performance, match involvement, and consequently increased effort in official matches, might be explained, at least in part by the modulation based on the above-mentioned mechanisms. Additionally, considering the M1-DLPFC montage, the issue of pain perception and the pain matrix appear to be fundamental for explaining the effects. Pain perception is controlled by a complex network of brain regions and circuits, known as the pain matrix (neuromatrix), which involves cognitive, emotional, and affective components. The DLPFC and the medial prefrontal cortex (mPFC) are important areas involved in pain perception. These regions also contribute to the cognitive processing of the painful experience, especially in aspects related to pain prediction, evaluation, and reinterpretation. Neuroimaging studies suggest that the reduction in pain levels after DLPFC stimulation may be related to the connectivity between this region and other pain perception areas, such as the cingulate cortex, insula, amygdala, and thalamus41.
There is evidence that painful stimuli produce an activation pattern in the DLPFC, suggesting an essential role for this region in the interpretation of the painful stimulus. In chronic pain conditions, neuroplasticity appears to produce an imbalance in the allocation of attentional and cognitive resources, leading to a distorted perception of pain23. The DLPFC, therefore, seems to play a central role in this imbalance. Thus, excitatory stimulation of the DLPFC could inhibit this dysfunctional cognitive and attentional allocation, leading to pain reduction. This rebalancing as a response to tDCS treatment mediated by the DLPFC involves a central issue regarding pain: because pain is subjective and has an emotional element, it will not always be directly related to the magnitude of the nociceptive signal31,50. Therefore, with tissue damage and chronic pain perception, neural plasticity mechanisms—the focus of tDCS intervention in pain treatment—may lead the athlete to develop greater pain perception than the actual damage, due to neuroplasticity-related adaptations.
A critical concern in sports medicine is whether neuromodulatory analgesia might "mask" pain, leading the athlete to overload a compromised structure and risk a complete rupture. However, our findings suggest that the tDCS-mediated pain reduction occurred in parallel with, and perhaps facilitated, structural tissue repair.
The multi-sequence MRI identified intermediate-signal intensity tissue filling the previous defect. This radiological finding is significant; it indicates the transition from an acute, fluid-filled tear to organized fibroblastic scar formation. The mechanism behind this might be explained by the emerging understanding of tDCS-mediated neuro-immune crosstalk. Studies have demonstrated that tDCS can promote a local anti-inflammatory environment by reducing lymphocyte infiltration and neovascularization while modulating systemic cytokine levels51. Specifically, the suppression of pro-inflammatory mediators such as TNF-alpha, IL-1beta, and IL-6, combined with an increase in the anti-inflammatory cytokine IL-10, likely creates a biological milieu conducive to tissue repair. Furthermore, it could be hypothesized that in the current reported case study, the restructuring evoked by tDCS in conjunction with the described multimodal rehabilitation program might induce modification of the dysfunctional cognitive allocation of attention, bringing the perception of pain closer to the reality of the healing damage. Rather than a dangerous "masking" effect, the intervention appears to have facilitated optimal loading, the controlled mechanical stress necessary for tendon remodeling, without exceeding the structural limits of the healing fibers.
5. Study limitations
While the results of this case report are promising, several limitations must be acknowledged to provide a balanced interpretation of the findings: as a single-case study (n=1), the results cannot be statistically generalized to the broader population of professional soccer players or different types of tendon injuries. The specific physiological profile and baseline physical conditioning of an elite athlete may influence the efficacy of tDCS differently than in a sedentary or recreational population. The athlete underwent a multifaceted rehabilitation program, including physical therapy and controlled workload management. Although the dual-site tDCS was the primary neuromodulatory addition, it is difficult to isolate its specific contribution to structural healing versus the natural history of tendon repair and standard-of-care physiotherapy. Indeed, without a sham-tDCS control, the "placebo effect" or "Hawthorne effect", common in high-performance environments where athletes are highly motivated to return to play, cannot be entirely ruled out. The athlete’s belief in the technology may have contributed to the observed reduction in VAS scores. While we hypothesize that the M1-DLPFC montage improved motor efficiency and activated the descending pain modulatory system (DPMS), we did not utilize functional MRI (fMRI) or Transcranial Magnetic Stimulation (TMS) to measure actual changes in cortical excitability or connectivity during the protocol. Despite these limitations, this case provides a unique 'proof-of-concept' for the safe integration of tDCS in elite sports, demonstrating that neuromodulation can coexist with structural tissue repair without masking symptoms to the point of reinjury.
6. Final considerations and conclusion
The longitudinal data from this case study demonstrates a positive clinical trajectory characterized by a significant reduction in Achilles’ tendon pain, stable perceived exertion during recovery, and a heightened capacity for competitive output. This "pain-performance paradox", where the athlete increased match intensity (RPE) while experiencing declining pain (VAS) suggests that the dual-site tDCS (M1-DLPFC) protocol effectively optimized the athlete’s performance. Critically, the objective structural recovery confirmed via serial ultrasonography and multi-sequence MRI indicates that this neuromodulatory intervention did not merely "mask" nociceptive signaling. Instead, it likely facilitated an optimal environment for tissue repair by modulating the top-down inhibitory pathways and potentially dampening systemic pro-inflammatory markers. The current findings suggest that tDCS is a promising, non-invasive adjunct for the management of tendon-related pain in elite athletes. When integrated into a multidisciplinary sports medicine framework, it may allow for the maintenance of high-level performance during the structural healing phase of an injury. However, while these results are encouraging, future research involving larger cohorts and sham-controlled designs is essential to further elucidate the specific mechanisms of tDCS on tendon-tissue remodeling and to standardize protocols for the professional athletic population.
Funding
This research received no specific grant from public, commercial, or not-for-profit funding agencies. A.M. is supported by CNPq (304269/2021-2); A.M and L.A.M. were supported by The São Paulo Research Foundation - Research, Innovation and Dissemination Centers - CEPID/BRAINN - The Brazilian Institute of Neuroscience and Neurotechnology (FAPESP Process: 13/07559-3).
Authors’ contributions
The authors declared that they have made substantial contributions to the work in terms of the conception or design of the research; the acquisition, analysis or interpretation of data for the work; and the writing or critical review for relevant intellectual content. All authors approved the final version to be published and agreed to take public responsibility for all aspects of the study.
Competing interests
No financial, legal, or political conflicts involving third parties (government, private companies, and foundations, etc.) were declared for any aspect of the submitted work (including but not limited to grants and funding, advisory board participation, study design, manuscript preparation, statistical analysis, etc.).
References
1. Nitsche MA, Cohen LG, Wassermann EM, Priori A, Lang N, Antal A, et al. Transcranial direct current stimulation: state of the art 2008. Brain Stimul. 2008;1(3):206-23. https://doi.org/10.1016/j.brs.2008.06.004
2. Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 2000;527(Pt 3):633-9. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00633.x
3. Naegel S, Biermann J, Theysohn N, Kleinschnitz C, Diener HC, Katsarava Z, et al. Polarity-specific modulation of pain processing by transcranial direct current stimulation – a blinded longitudinal fMRI study. J Headache Pain. 2018;19(1):99. https://doi.org/10.1186/s10194-018-0924-5
4. Lang N, Siebner HR, Ward NS, Lee L, Nitsche MA, Paulus W, et al. How does transcranial DC stimulation of the primary motor cortex alter regional neuronal activity in the human brain? Eur J Neurosci. 2005;22(2):495-504. https://doi.org/10.1111/j.1460-9568.2005.04233.x
5. Bahr-Hosseini M, Bikson M. Neurovascular-modulation: a review of primary vascular responses to transcranial electrical stimulation as a mechanism of action. Brain Stimul. 2021;14(4):837-47. https://doi.org/10.1016/j.brs.2021.04.015
6. Gellner AK, Frase S, Reis J, Fritsch B. Direct current stimulation increases blood flow and permeability of cortical microvasculature in vivo. Eur J Neurol. 2023;30(2):362-71. https://doi.org/10.1111/ene.15616
7. Souza L, Moscaleski LA, Fonseca A, Fernandes VG, Nery GM, Morya E, et al. The competition between brain and body: does performing simultaneous cognitive and physical tasks alter the cortical activity of athletes compared to performing these tasks in isolation? Physiol Behav. 2025;297:114936. https://doi.org/10.1016/j.physbeh.2025.114936
8. Fernandes VG, Moscaleski LA, Fonseca A, Morya E, Okano AH, Arruda AFS, et al. The dynamics of brain frequency bands and response to neuromodulation during cognitive testing in rugby sevens athletes throughout a sports season. Curr Psychol. 2024;43(39):30629-43. https://doi.org/10.1007/s12144-024-06682-2
9. Gonçalves DS, Moscaleski LA, Silva GM, Morgans R, Okano AH, Moreira A. The effect of combined transcranial direct current stimulation and pneumatic compression as part of a comprehensive recovery strategy in professional male top-level soccer players. J Strength Cond Res. 2024;38(9):1658-66. https://doi.org/10.1519/JSC.0000000000004844
10. Mehrsafar AH, Rosa MAS, Zadeh AM, Gazerani P. A feasibility study of application and potential effects of a single session transcranial direct current stimulation (tDCS) on competitive anxiety, mood state, salivary levels of cortisol and alpha amylase in elite athletes under a real-world competition. Physiol Behav. 2020;227:113173. https://doi.org/10.1016/j.physbeh.2020.113173
11. Moreira A, Machado DGS, Moscaleski L, Bikson M, Unal G, Bradley PS, et al. Effect of tDCS on well-being and autonomic function in professional male players after official soccer matches. Physiol Behav. 2021;233:113351. https://doi.org/10.1016/j.physbeh.2021.113351
12. Moreira A, Machado DGS, Bikson M, Unal G, Bradley PS, Moscaleski L, et al. Effect of transcranial direct current stimulation on professional female soccer players' recovery following official matches. Percept Mot Skills. 2021;128(4):1504-29. https://doi.org/10.1177/00315125211021239
13. Moreira A, Machado DGS, Moscaleski LA, Baptista AF, Li LM, Morya E, et al. tDCS in exercise, sport performance, and recovery process. In: Brunoni AR, Nitsche MA, Loo CK. Transcranial Direct Current Stimulation in Neuropsychiatric Disorders. 2nd ed. Switzerland: Springer; 2021. p. 413-32. https://doi.org/10.1007/978-3-030-76136-3_18
14. Moreira A, Moscaleski L, Machado DGS, ikson M, Unal G, Bradley PS, et al. Transcranial direct current stimulation during a prolonged cognitive task: the effect on cognitive and shooting performances in professional female basketball players. Ergonomics. 2023;66(4):492-505. https://doi.org/10.1080/00140139.2022.2096262
15. Moscaleski LA, Fonseca A, Brito R, Morya E, Morgans R, Moreira A, et al. Does high-definition transcranial direct current stimulation change brain electrical activity in professional female basketball players during free-throw shooting? Front Neuroergon. 2022;3:932542. https://doi.org/10.3389/fnrgo.2022.932542
16. Okano AH, Fontes EB, Montenegro RA, Farinatti PTV, Cyrino ES, Li LM, et al. Brain stimulation modulates the autonomic nervous system, rating of perceived exertion and performance during maximal exercise. Br J Sports Med. 2015;49(18):1213-8. https://doi.org/10.1136/bjsports-2012-091658
17. Valenzuela PL, Amo C, Sánchez-Martínez G, Torrontegi E, Vázquez-Carrión J, Montalvo Z, et al. Enhancement of mood but not performance in elite athletes with transcranial direct-current stimulation. Int J Sports Physiol Perform. 2019;14(3):310-6. https://doi.org/10.1123/ijspp.2018-0473
18. Flood A, Waddington G, Cathcart S. High-definition transcranial direct current stimulation enhances conditioned pain modulation in healthy volunteers: a randomized trial. J Pain. 2016;17(5):600-5. https://doi.org/10.1016/j.jpain.2016.01.472
19. Boggio PS, Zaghi S, Lopes M, Fregni F. Modulatory effects of anodal transcranial direct current stimulation on perception and pain thresholds in healthy volunteers. Eur J Neurol. 2008;15(10):1124-30. https://doi.org/10.1111/j.1468-1331.2008.02270.x
20. Flood A, Waddington G, Keegan RJ, Thompson KG, Cathcart S. The effects of elevated pain inhibition on endurance exercise performance. PeerJ. 2017;5:e3028. https://doi.org/10.7717/peerj.3028
21. Rêgo GG, Lapenta OM, Marques LM, Costa TL, Leite J, Carvalho S, et al. Hemispheric dorsolateral prefrontal cortex lateralization in the regulation of empathy for pain. Neurosci Lett. 2015;594:12-6. https://doi.org/10.1016/j.neulet.2015.03.042
22. Mariano TY, Wout MV, Garnaat SL, Rasmussen SA, Greenberg BD. Transcranial direct current stimulation (tDCS) targeting left dorsolateral prefrontal cortex modulates task-induced acute pain in healthy volunteers. Pain Med. 2016;17(4):737-45. https://doi.org/10.1093/pm/pnv042
23. Vaseghi B, Zoghi M, Jaberzadeh S. The effects of anodal-tDCS on corticospinal excitability enhancement and its after-effects: conventional vs. unihemispheric concurrent dual-site stimulation. Front Hum Neurosci. 2015;9:533. https://doi.org/10.3389/fnhum.2015.00533
24. Ong WY, Stohler CS, Herr DR. Role of the prefrontal cortex in pain processing. Mol Neurobiol. 2019;56(2):1137-66. https://doi.org/10.1007/s12035-018-1130-9
25. Sankarasubramanian V, Cunningham DA, Potter-Baker KA, Beall EB, Roelle SM, Varnerin NM, et al. Transcranial direct current stimulation targeting primary motor versus dorsolateral prefrontal cortices: proof-of-concept study investigating functional connectivity of thalamocortical networks specific to sensory-affective information processing. Brain Connect. 2017;7(3):182-96. https://doi.org/10.1089/brain.2016.0440
26. McKinney J, Velghe J, Fee J, Isserow S, Drezner JA. Defining athletes and exercisers. Am J Cardiol. 2019;123(3):532-5. https://doi.org/10.1016/j.amjcard.2018.11.001
27. Hawker GA, Mian S, Kendzerska T, French M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res (Hoboken). 2011;63(suppl 11):S240-52. https://doi.org/10.1002/acr.20543
28. Foster C, Florhaug JA, Franklin J, Gottschall L, Hrovatin LA, Parker S, et al. A new approach to monitoring exercise training. J Strength Cond Res. 2001;15(1):109-15. Cited: PMID: 11708692
29. Korats G, Le Cam S, Ranta R, Hamid M. Applying ICA in EEG: choice of the window length and of the decorrelation method. In: Gabriel J, Schier J, Huffel S, Conchon E, Correia C, Fred A, et al. Biomedical Engineering Systems and Technologies. Vol 357. Berlin: Springer; 2013. p. 269-86. https://doi.org/10.1007/978-3-642-38256-7_18
30. Seibt O, Truong D, Khadka N, Huang Y, Bikson M. Computational finite element method (FEM) forward modeling workflow for transcranial direct current stimulation (tDCS) current flow on MRI-derived head: Simpleware and COMSOL Multiphysics tutorial [preprint]. bioRxiv. 2019. https://doi.org/10.1101/704940
31. Rainville P. Brain mechanisms of pain affect and pain modulation. Curr Opin Neurobiol. 2002;12(2):195-204. https://doi.org/10.1016/S0959-4388(02)00313-6
32. Bornhövd K, Quante M, Glauche V, Bromm B, Weiller C, Büchel C. Painful stimuli evoke different stimulus-response functions in the amygdala, prefrontal, insula and somatosensory cortex: a single-trial fMRI study. Brain. 2002;125(Pt 6):1326-36. https://doi.org/10.1093/brain/awf137
33. Nascimento RM, Cavalcanti RL, Souza CG, Chaves G, Macedo LB. Transcranial direct current stimulation combined with peripheral stimulation in chronic pain: a systematic review and meta-analysis. Expert Rev Med Devices. 2023;20(2):121–40. https://doi.org/10.1080/17434440.2022.2039623
34. Vitor-Costa M, Okuno NM, Bortolotti H, Bertollo M, Boggio PS, Fregni F, et al. Improving Cycling Performance: Transcranial Direct Current Stimulation Increases Time to Exhaustion in Cycling. PLoS One. 2015;10(12):e0144916. https://doi.org/10.1371/journal.pone.0144916
35. Laurent B, Peyron R, Larrea LG, Mauguière F. Positron emission tomography to study central pain integration. Rev Neurol [Internet]. 2000;156(4):341-51. Available from: https://pubmed.ncbi.nlm.nih.gov/10795010/
36. Mouraux A, Diukova A, Lee MC, Wise RG, Iannetti GD. A multisensory investigation of the functional significance of the "pain matrix". Neuroimage. 2011;54(3):2237-49. https://doi.org/10.1016/j.neuroimage.2010.09.084
37. Antal A, Terney D, Kühnl S, Paulus W. Anodal transcranial direct current stimulation of the motor cortex ameliorates chronic pain and reduces short intracortical inhibition. J Pain Symptom Manage. 2010;39(5):890-903. https://doi.org/10.1016/j.jpainsymman.2009.09.023
38. Antal A, Brepohl N, Poreisz C, Boros K, Csifcsak G, Paulus W. Transcranial direct current stimulation over somatosensory cortex decreases experimentally induced acute pain perception. Clin J Pain. 2008;24(1):56-63. https://doi.org/10.1097/AJP.0b013e318157233b
39. Grundmann L, Rolke R, Nitsche MA, Pavlakovic G, Happe S, Treede RD, et al. Effects of transcranial direct current stimulation of the primary sensory cortex on somatosensory perception. Brain Stimul. 2011;4(4):253-60. https://doi.org/10.1016/j.brs.2010.12.002
40. Bingel U, Quante M, Knab R, Bromm B, Weiller C, Büchel C. Single trial fMRI reveals significant contralateral bias in responses to laser pain within thalamus and somatosensory cortices. Neuroimage. 2003;18(3):740-8. https://doi.org/10.1016/S1053-8119(02)00033-2
41. Porro CA. Functional imaging and pain: behavior, perception, and modulation. Neuroscientist. 2003;9(5):354-69. https://doi.org/10.1177/1073858403253660
42. Coghill RC, Sang CN, Maisog JM, Iadarola MJ. Pain intensity processing within the human brain: a bilateral, distributed mechanism. J Neurophysiol. 1999;82(4):1934-43. Cited: PMID: 10515983
43. Fomberstein K, Qadri S, Ramani R. Functional MRI and pain. Curr Opin Anaesthesiol. 2013;26(5):588-93. https://doi.org/10.1097/01.aco.0000433060.59939.fe
44. Apkarian AV, Bushnell MC, Treede RD, Zubieta JK. Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain. 2005;9(4):463-84. https://doi.org/10.1016/j.ejpain.2004.11.001
45. Baliki MN, Petre B, Torbey S, Herrmann KM, Huang L, Schnitzer TJ, et al. Corticostriatal functional connectivity predicts transition to chronic back pain. Nat Neurosci. 2012;15(8):1117-9. https://doi.org/10.1038/nn.3153
46. Sawamoto N, Honda M, Okada T, Hanakawa T, Kanda M, Fukuyama H, et al. Expectation of pain enhances responses to nonpainful somatosensory stimulation in the anterior cingulate cortex and parietal operculum/posterior insula: an event-related functional magnetic resonance imaging study. J Neurosci. 2000;20(19):7438-45. https://doi.org/10.1523/JNEUROSCI.20-19-07438.2000
47. Ploghaus A, Tracey I, Gati JS, Clare S, Menon RS, Matthews PM, et al. Dissociating pain from its anticipation in the human brain. Science. 1999;284(5422):1979-81. https://doi.org/10.1126/science.284.5422.1979
48. Bolognini N, Pascual-Leone A, Fregni F. Using non-invasive brain stimulation to augment motor training-induced plasticity. J Neuroeng Rehabil. 2009;6:8. https://doi.org/10.1186/1743-0003-6-8
49. Liebetanz D, Nitsche MA, Tergau F, Paulus W. Pharmacological approach to the mechanisms of transcranial DC-stimulation-induced after-effects of human motor cortex excitability. Brain. 2002;125(Pt 10):2238-47. https://doi.org/10.1093/brain/awf238
50. Mauger AR, Jones AM, Williams CA. Influence of acetaminophen on performance during time trial cycling. J Appl Physiol. 2010;108(1):98-104. Cited: PMID: 19910336
51. Zancanaro M, Stein DJ, Lopes BC, Souza A, Toledo RS, Souza AH, et al. Preemptive transcranial direct current stimulation induces analgesia, prevents chronic inflammation and fibrosis, and promotes tissue repair in a rat model of postoperative pain. Neurosci Lett. 2023;813:137407. https://doi.org/10.1016/j.neulet.2023.137407